|
of metal surfaces used in food processing
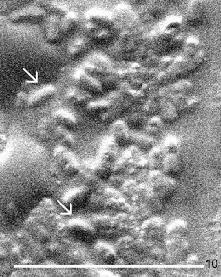
Most foods consumed in industrially developed countries are made on an industrial scale. Stainless steel is the material from which most of the processing equipment is made. It is sturdy, resistant to wear and corrosion, and relatively easy to clean. Smooth surface finish is essential to cleanability. Contaminants adhere more readily and firmly to a rough than to a smooth surface and they may be much more difficult to remove from areas which are not easily or not at all accessible to mechanical cleaning. Regular cleaning is very important in order not to allow microorganisms to propagate and to spoil the foods being processed. Meat, milk, dough, cooked vegetables and other foods provide environments which are rapidly occupied by a wide variety of microorganisms, many of which may cause food poisoning. Food processing surfaces may appear clean to a naked eye and yet there may be bacteria attached to them. Bacteria are small, approximately 1 µm in diameter, and, thus, may easily be hiding in minute scratches. Their presence may be revealed by washing them into a nutrient medium and culturing them whereby their identity may also be revealed. However, many bacteria firmly adhere to various surfaces and would not be transferred easily. It may be of interest to see them attached to the surface. In addition, it may also be of interest to examine the food processing surface in greater detail using electron microscopy. Since it is not feasible to place large metal pieces into an electron microscope, the best approach is to replicate the surface and examine the replica. Stainless steel squares, 40 mm x 40 mm, were used in the experiments. Smaller squares, about 5 mm x 5 mm were inscribed in the steel and numbered using a sharp file. Then the steel was cleaned with acetone and its surface was replicated at various squares. For each experiment, 3 squares, 10 mm x 10 mm, of replicating tape No. 44840 were used. The area to be replicated was wetted with acetone and the first replicating tape piece was laid over it to avoid the formation of any bubbles. This tape was again wetted with acetone and the second piece of the tape was applied. Using the same procedure, the third piece was used and the acetone was allowed to evaporate. When the tape was completely dry, it was peeled off and mounted on double sticky tape attached to 13 mm SEM stubs. The replicas were then coated with gold and examined at 3 kV in a Philips XL30 ESEM (environmental scanning electron microscope). The steel disks were then coated with gold and also examined by the same electron microscope in order to compare images of the original surface with those of the replicas. Since harmless food-borne bacteria were easily available in a fermented vegetable salad, a few drops of their suspensions were placed on another steel square and allowed to dry. Areas with the dried bacteria were replicated using the same procedure as before. The steel squares were then also gold-coated and examined by SEM. A good correlation was found between the images of clean steel squares and the replicas, where the numbers inscribed by a file made the comparison relatively easy. It was, however, almost impossible to find the corresponding areas on the steel disks and in the replicas without the aid of a marker. One of the problems was the presence of other solids such as salt in the bacterial suspension. Furthermore, the "inversion" of the images - convex areas on the steel appeared as concave areas in the replica - made orientation to find the same area on the monitor was very difficult. Bacteria dried on the steel disks were easy to examine directly following their gold-coating. However, their drying with the other solids in air without fixation, dehydration and CPD has led to considerably poorer images (top pair of micrographs) than those of bacteria properly prepared for SEM. The replicating tape made it possible to photograph bacteria in the contaminated areas (bottom pair of micrographs) in spite of difficulties associated with inverted images and a complex mix of additional components. The "dimples" in the left image are actually bacteria attached to the steel surface. Inverting the signal in Adobe Photoshop produced the image shown at right thus making it clear that there were bacteria in the area captured by the replica. | ||||||||
The top and bottom pairs of micrographs were photographed at 3000x and 4000x magnifications, respectively. Interestingly, the bacteria present in the dried marinate have not been absorbed into the replicating tape but provided imprints. This study is being expanded to other metal and also acetone-resistant plastic surfaces.
Links to related subjects:
Stainless steel equipment is common in food processing plants because it has many beneficial properties such as
sturdiness, resistance to physical as well as chemical damage and thus
is easy to clean. An example of criteria for newly constructed or renovated food establishments has been published by the Marion County Health Department, Indianapolis, Indiana, USA.
Properties of various stainless steel types including their resistance to the foods and cleaning agents with which they may come into contact are described in New Guidance Documents Governing the Selection and Safety-Evaluation of Materials for Food Use.
Specific demands of sanitation in the manufacture of various food are discussed to in several articles, such as: Microorganisms are too small to be seen by a naked eye. They may easily adhere to rough surfaces and not be detected except by their ability to grow if the environment is suitable. Assessment of the fine structure of the surface may be accomplished by scanning electron microscopy but it is not possible to take small samples of the food processing surfaces. One of the procedures, which would allow visualization of such surfaces, is their replication. There are reports on the replication of metal surfaces for SEM, such as:
Bacteria are everywhere. How do we keep them out of our food? Acknowledgment | ||||||||
| Updated January 25, 2005 | ||||||||
© SCIMAT 2005